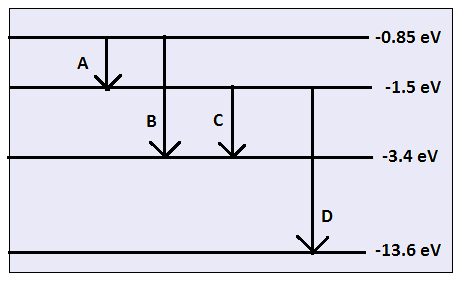
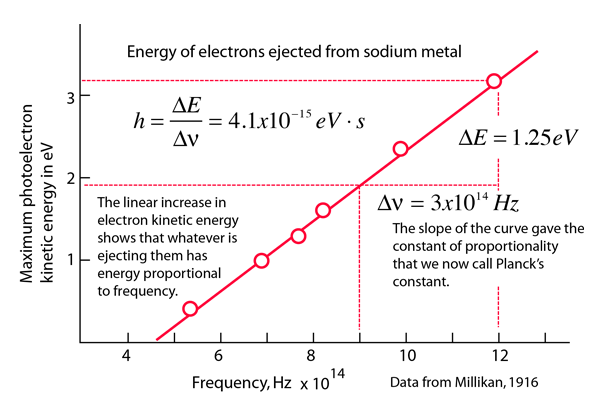
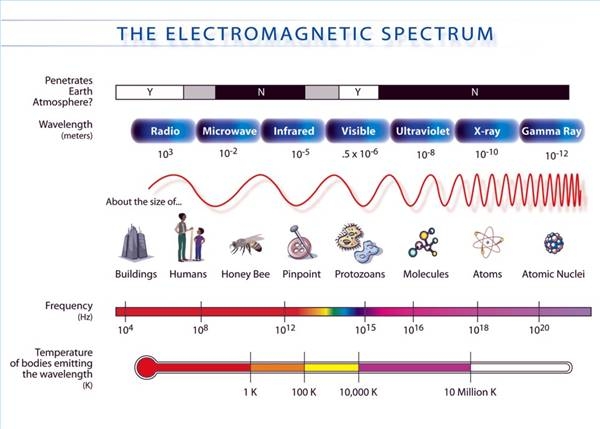
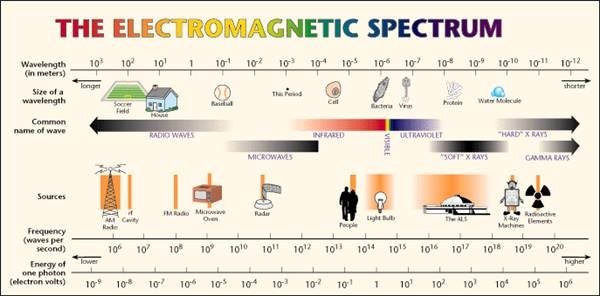
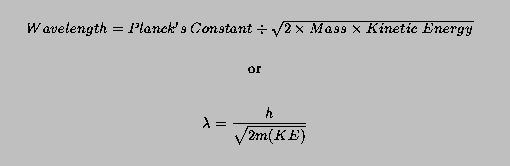Calculating particle properties of a wave Ch. 12 A light wave consists of particles (photons): The energy E of the particle is calculated from the frequency. - ppt download

Question Video: Identifying an Electron Energy Level Transition Given the Wavelength of an Absorbed Photon | Nagwa

The maximum wavelength of light photoelectric effect from a metal is 200 nm. The maximum kinetic energy of electron which is emitted by the radiation of wave length 100 nm will be:

Question Video: Understanding a Photon Emitted By an Electron Transitioning From Energy Level 3 to 2 | Nagwa
The work function of a metal is 10.0 eV. The longest wavelength of light that can cause photoelectron emission from this metal is approximately Options: a 248 nm b 124 nm b