Table 1 from Effects of solvent polarity and solvent viscosity on the fluorescent properties of molecular rotors and related probes. | Semantic Scholar

Enhancing Metal Separations by Liquid–Liquid Extraction Using Polar Solvents - Li - 2019 - Chemistry – A European Journal - Wiley Online Library
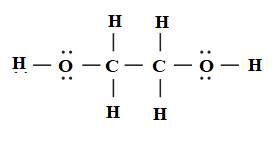
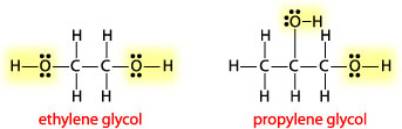
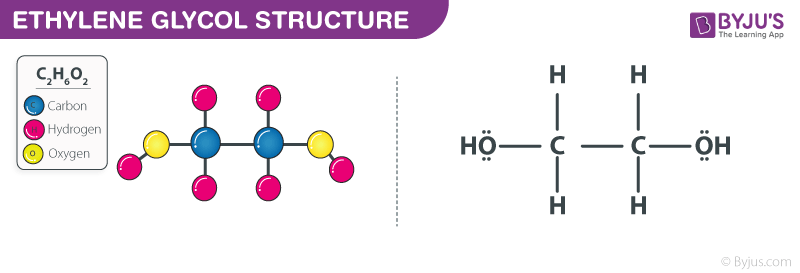
The Lewis structure of ethylene glycol is to be drawn. The sigma and pi bonds in ethylene glycol are to be identified. Concept introduction: The strategy for drawing Lewis structure is mention

Is Ethylene Glycol Polar or Nonpolar? – (Polarity of C2H6O2) | Ethylene glycol, Polar, Molecular geometry

a) MilliQ water and ethylene glycol contact angles of GGM-L films; (b)... | Download Scientific Diagram
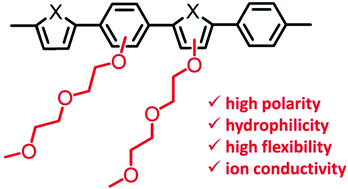
Oligo(ethylene glycol) as side chains of conjugated polymers for optoelectronic applications - Polymer Chemistry (RSC Publishing)

Synthesis and Nanoencapsulation of Poly(ethylene glycol)-Distearates Phase Change Materials for Latent Heat Storage and Release | ACS Applied Energy Materials

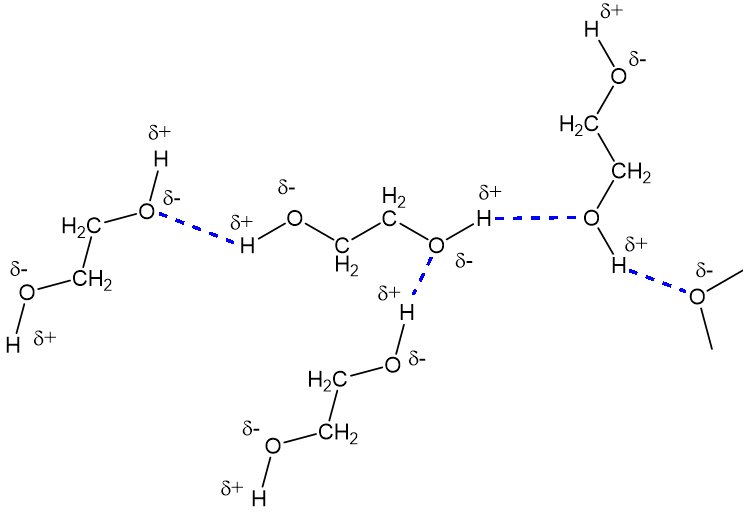
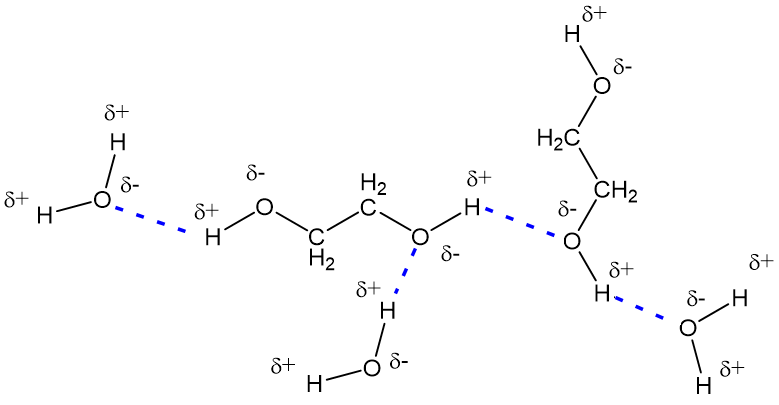
Ethylene glycol, HOCH_2CH_2OH, may look nonpolar when drawn, but an internal hydrogen bond results in an electric dipole moment. Explain. | Homework.Study.com
![PDF] Ketoprofen Solubility in Organic Solvents and Aqueous Co-solvent Systems : Interactions and Thermodynamic Parameters of Solvation | Semantic Scholar PDF] Ketoprofen Solubility in Organic Solvents and Aqueous Co-solvent Systems : Interactions and Thermodynamic Parameters of Solvation | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/0b800835ae11aba75ea4974e9f8d2f1041157737/4-Table1-1.png)
PDF] Ketoprofen Solubility in Organic Solvents and Aqueous Co-solvent Systems : Interactions and Thermodynamic Parameters of Solvation | Semantic Scholar
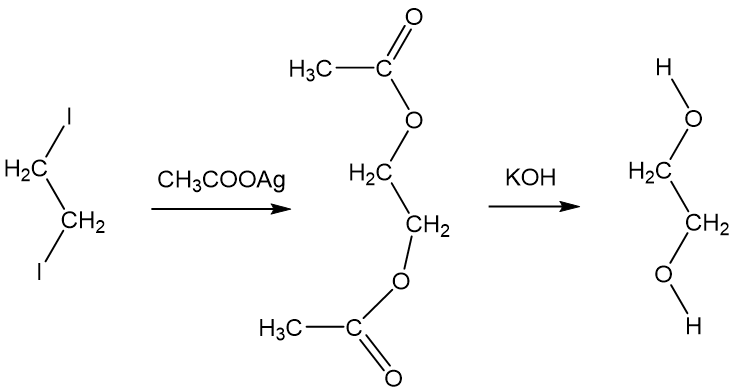
Ethylene glycol is liquid at room temperature and soluble in water in all proportions. n-butane, CH3CH2CH2 CH2CH3, is gas at room temp and insoluble in water. How do you account for these